Medicare to Test New Payment Models for Outpatient Drugs
Progress on policies to slow the growth of health care spending is expected to be slow in an election year, but this week, the Center for Medicare and Medicaid Services (CMS) announced a proposed rule to test new ways to pay providers or charge beneficiaries for outpatient drugs in Medicare Part B, including testing a proposal we put forward in our PREP Plan last year. These payment model tests, which will be run by the Center for Medicare and Medicaid Innovation (CMMI) created in the Affordable Care Act, intend to encourage more efficient and effective use of outpatient drugs by correcting some of the flawed incentives in the current system. This announcement is a step forward in encouraging more efficient health care delivery, and hopefully at least some of these strategies will prove useful enough to be expanded on and produce significant budgetary savings in future years.
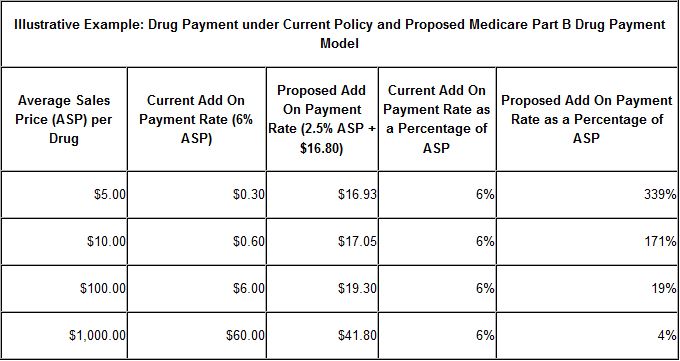
The tests will come in two steps. The first step involves changing the formula for reimbursing physicians to administer outpatient drugs, which is currently set at the average sales price (ASP) of the drug plus a 6 percent add-on payment. This formula may encourage physicians to use more expensive drugs since the add-on payment grows with the cost. The new model that would be tested starting late 2016 would change the add-on payment to 2.5 percent of the ASP plus a flat fee of $16.80 (which would be indexed for inflation in future years). This would reduce the incentive to use more expensive drugs; for example, the difference in the add-on payment for a $5 drug and a $1,000 drug is currently $59.70 but would be reduced to $24.87 under the new formula.
This policy is similar to one we proposed in our PREP Plan, though our policy would have converted the entire add-on payment to a flat fee and would have saved $10 billion over ten years. It is also similar to an option the Medicare Payment Advisory Commission (MedPAC) evaluated, with them saying that "A flat-fee add-on may help address concerns about reimbursement for very inexpensive drugs." The CMS policy is intended to be budget-neutral on a static basis assuming no change in physician behavior, but it would produce savings if physicians used less expensive drugs in response to the change in incentives. An analysis of a proposal to convert the entire add-on payment to a flat fee found savings of $4 billion over ten years as a result of the shift to lower-cost drugs.
Source: CMS
The second step in these payment model tests would use a third party to test out value-based purchasing strategies. These strategies include:
- Reduced patient cost-sharing, to steer beneficiaries towards more cost-effective drugs which would involve some direct costs but would likely be more than offset by lower hospitalization costs.
- Evidence-based decision support tools to be a resource for providers on appropriate use of drugs.
- Indications-based pricing, which would vary payments for drugs based on their effectiveness in treating different conditions.
- Reference pricing, which would set one payment rate for therapeutically similar drugs.
- Risk-sharing agreements, where CMS would enter into voluntary agreements with drug companies to link prices and patient outcomes.
CMS will have four different test groups: one experiencing no change from current law, one experiencing only the ASP reimbursement change, one experiencing only the value-based purchasing changes, and one experiencing both sets of changes. The tests will run for five years.
Policymakers face tremendous challenges in limiting the growth of health care spending without sacrificing the quality of or access to necessary care. These policies have the potential to both improve quality of care and reduce costs by better aligning incentives away from the use of more costly than necessary drugs towards more effective treatment. These are the sort of tests that make the CMMI important to health care policy and the budget. It will be interesting to watch this demonstration progress and whether it will prove successful and be expanded.


